H2FC Technology
Fuel Cell Technology
Fuel cells generate electricity by an electrochemical reaction in which oxygen and a hydrogen-rich fuel combine to form water. Unlike internal combustion engines, the fuel is not combusted, the energy instead being released electrocatalytically. This allows fuel cells to be highly energy efficient, especially if the heat produced by the reaction is also harnessed for space heating, hot water or to drive refrigeration cycles.
There are several different types of fuel cell, but they are all based around a central design. A fuel cell unit consists of a stack, which is composed of several individual cells. Each cell within the stack has two electrodes, one positive and one negative, called the cathode and the anode. The reactions that produce electricity take place at the electrodes. Every fuel cell also has either a solid or a liquid electrolyte, which carries ions from one electrode to the other, and a catalyst, which accelerates the reactions at the electrodes. The electrolyte plays a key role – it must permit only the appropriate ions to pass between the electrodes. If free electrons or other substances travel through the electrolyte, they disrupt the chemical reaction and lower the efficiency of the cell.
Fuel cells are generally classified according to the nature of the electrolyte (except for direct methanol fuel cells which are named for their ability to use methanol as a fuel), each type requiring particular materials and fuel. Each fuel cell type also has its own operational characteristics, offering advantages to particular applications. This makes fuel cells a very versatile technology. As a result, fuel cells have a broader range of application than any other currently available power source – from toys to large power plants, from vehicles to mobile chargers, and from household power to battlefield power.
Main types of fuel cells: (1) PEMFC (proton exchange membrane fuel cells), (2) DMFC (direct methanol fuel cells), (3) SOFC (solid oxide fuel cells), (4) AFC (alkaline fuel cells), (5) MCFC (molten carbonate fuel cells), (6) PAFC (phosphoric acid fuel cells). (Source: http://www.fuelcelltoday.com)
Fuel Cell
A fuel cell is a device that uses hydrogen (or hydrogen-rich fuel) and oxygen to create electricity. Fuel cells are more energy-efficient than combustion engines and the hydrogen used to power them can come from a variety of sources. If pure hydrogen is used as a fuel, fuel cells emit only heat and water, eliminating concerns about air pollutants or greenhouse gases.
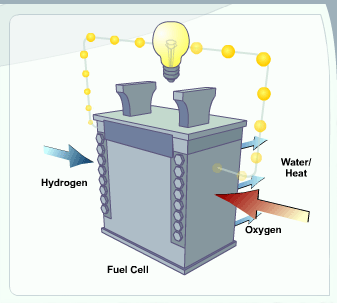
https://www.fueleconomy.gov/feg/fcv_PEM.shtml
PEM Fuel Cell

https://www.fueleconomy.gov/feg/fcv_PEM.shtml
Hydrogen Production
Once produced, hydrogen generates electrical power in a fuel cell, emitting only water vapor and warm air. It holds promise for growth in both the stationary and transportation energy sectors. Hydrogen can be produced from resources like natural gas, coal, solar energy, wind, and biomass. Hydrogen-powered fuel cell electric vehicles emit none of harmful substances. Their only emissions are water (H2O) and warm air. The environmental and health benefits are also seen at the source of hydrogen production if derived from low- or zero-emission sources, such as solar, wind, and nuclear energy and fossil fuels with advanced emission controls and carbon sequestration.
Hydrogen’s energy content by volume is low. This makes storing hydrogen a challenge because it requires high pressures, low temperatures, or chemical processes to be stored compactly. Overcoming this challenge is important for light-duty vehicles because they often have limited size and weight capacity for fuel storage. The storage capacity for hydrogen in light-duty vehicles should enable a driving range of more than 300 miles to meet consumer needs. Because hydrogen has a low volumetric energy density compared to gasoline, storing this much hydrogen on a vehicle currently requires a larger tank than what is equipped on most conventional vehicles. To be competitive in the marketplace, the cost of fuel cells will have to decrease substantially without compromising vehicle performance. The cost to build and maintain hydrogen stations also needs to decrease for the market to be able to support a hydrogen economy. (Source: https://www.afdc.energy.gov)

http://www.hydrogenambassadors.com/background/h2-laws-physics.php
Hydrogen-based Energy System

Hydrogen-based energy system aims at transforming the fossil-based energy system towards increasing use of local renewable energy sources and clean technology solutions in end-consuming. The hydrogen and fuel cell technology solutions, already available in the market, enable the implementation of clean energy production and end-consuming in industrial, residential, commercial business and service sectors and in transportation. This view of hydrogen economy or hydrogen society is sometimes argued as hydrogen romanticism. On the contrary, this clean technology developed and brought onto the market offers the solutions to transform the fossil-based energy system and to stop climate warming. This is an option for the actors in the global value chain of hydrogen and fuel cell technology sector. The future energy system utilizing hydrogen as an energy carrier is illustrated below (Source: Technology Roadmap, International Energy Agency, 2015).
H2FC Market
Global Market
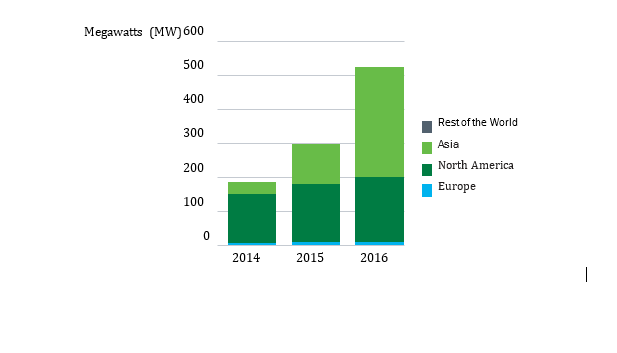
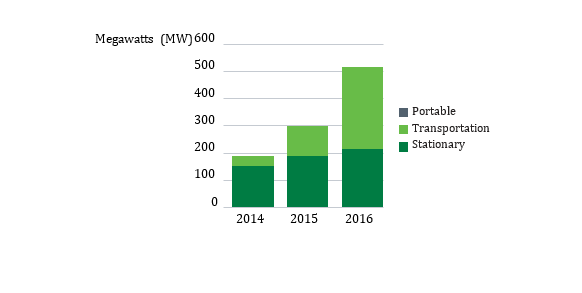

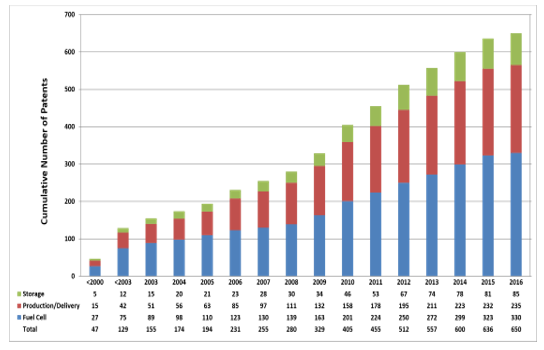
Patents
Top ten investors

Technology Development
- stationary power
- portable power
- auxiliary power units
- backup power
- material handling equipment
Thus, they emphasize strongly in they programs further research and development of applications, in particular, focusing on durability, performance and cost reductions.
Hydrogen-Economy

EU Targets
- 2020 sufficient recharging stations to allow electric cars to travel around densely populated areas within the network the country has determined;
- 2025 (end) sufficient recharging stations for hydrogen (for any country that decides to include hydrogen in its national policy framework);
- 2025 (end) sufficient liquefied natural gas (LNG) stations at seaports, to accommodate LNG-powered ships. Source: http://eurlex.europa.eu/summary/chapter/energy.html?root_default=SUM_1_CODED%3D18&locale=en